1. Introduction
We urge the National Pharmaceutical Regulatory Agency (NPRA) to withdraw the proposed Guidelines on Implementation of Patent Linkage for Pharmaceutical Products in Malaysia and to undertake meaningful consultations with civil society organizations and patient advocacy organisations on the best way to approach implementation of Article 18.53 of the Comprehensive and Progressive Agreement for Trans-Pacific Partnership (CPTPP).
The proposed Guidelines are CPTPP-plus going well beyond what is required under Article 18.53 of the CPTPP and will have serious adverse consequences for both the generic industry and access to affordable pharmaceutical products in Malaysia.
High medicine prices are a persistent challenge in Malaysia placing a significant burden on both patients and the public healthcare system. As a result, patients face high out-of-pocket costs, while the government must allocate substantial resources to procure medicines, constraining public health spending. Thus, it is absolutely critical that NPRA's implementation of Article 18.53 of the CPTPP does not erect barriers to the early entry of generic competition. Early generic competition is essential to reducing prices and improving access, and any regulatory approach that delays it will only exacerbate existing affordability challenges.
Looking at 10 years of wholesale and retail prices of medicines in Malaysia (2012-2022) the trend is incremental price increase (median increase: 40% and 33.6%), or stable increase (median increase: 15% and 16%), but none showed a decrease. In fact, the recent 5-year trend shows that 66% of wholesale and 68% of retail medicines have become more expensive. The increase for wholesale and retail prices is highly correlated.
The Ministry of Health’s 2024 pooled procurement data shows that a switch from innovator (patented) to generic medicines produced significant savings for the Ministry: in all cases, the saving was averaged at RM6.8mil per medicine procurement. However, when the Ministry sticks to the same innovator medicine (and not a generic), there is a 1 in 4 chance that the Ministry must fork out more for the same procurement. In the other 75% of cases the savings the Ministry could gain per procurement of originator medicines is about 12% on average after price negotiations or bulk buying. This is small compared to the savings made by a switch to generics. Hence any delay in availability of a generic pharmaceutical product in the market has a huge cost to the government and the public. Timely registration of a generic product by NPRA is therefore crucial, together with a generic importer or local manufacturer that is ready to enter the market with the registration in hand.
In 2024, the Ministry of Health (MOH) spent RM823 million on 839 innovator medicines, of which 99% were imported. Even a modest delay in the entry of generics—affecting just 5% of these medicines—will have significant financial implications. Based on estimated switch savings, a delay of just 5% of generics could result in loss of savings of approximately RM285.6 million, equivalent to about 8.2% of MOH’s total annual expenditure on medicine procurement.
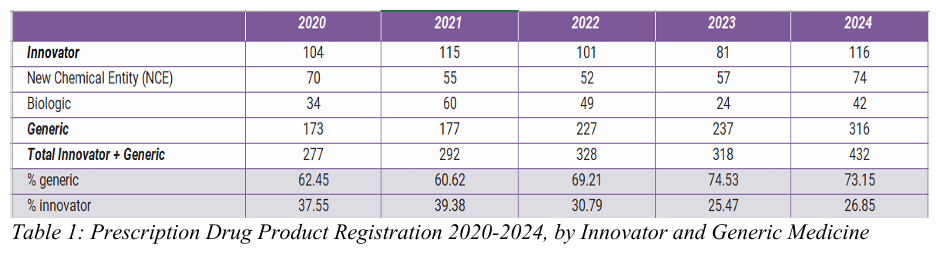 Over the five-year period between 2020 and 2024, the number and percentage of generic products approved by the NPRA has increased (See Table 1). In 2024, 316 generic prescription drugs were registered, accounting for 73.15% of the total. The progress and gains made under the National Generic Medicines Framework, which was developed based on the Malaysian National Medicines Policy and favours the use of generic medicines, will be reversed if the proposed Guidelines are adopted.
Over the five-year period between 2020 and 2024, the number and percentage of generic products approved by the NPRA has increased (See Table 1). In 2024, 316 generic prescription drugs were registered, accounting for 73.15% of the total. The progress and gains made under the National Generic Medicines Framework, which was developed based on the Malaysian National Medicines Policy and favours the use of generic medicines, will be reversed if the proposed Guidelines are adopted.
The Malaysian government has also identified the generic pharmaceutical industry as a strategic priority under the New Industrial Master Plan (NIMP) 2030. According to the NIMP, the Malaysian generic industry has grown significantly over the past decade, reaching 10 per cent growth of total local medicine sales between 2010 and 2021. The Plan sets out an ambition for Malaysia to maintain its position as a first mover in generics while expanding export capacity. More broadly, it envisions strengthening the industry to improve the affordability and accessibility of medicines, enhance national self-sufficiency, support local economic development, and reduce the pharmaceutical trade deficit. The NIMP pharmaceutical sector plan (2023) identifies evergreening patents as “significant challenge” of patents: “In Malaysia, the Patent Act does not exclude second medical use patent for pharmaceutical products. This can lead to monopolies and high prices for essential medicines. … Some companies may patent a new formulation with slight modifications of existing drugs. This allows the companies to extend its monopoly on the products and prevent generic competition.”
In this context, the proposed Guidelines are deeply concerning. In particular, the proposed Guidelines: (i) are CPTPP-plus; (ii) legitimises and promotes patent evergreening; (iii) its proposed patent listing is open to abuse; (iv) will encourage frivolous litigation against generic companies and create a chilling effect on generic market entry; (v) undermines the effective use of flexibilities in the WTO Agreement on Trade-related Aspects of Intellectual Property Rights (TRIPS); (vi) violates established principles of law; (vii) exceeds NPRA’s legal mandate and (viii) introduces legal uncertainty. These concerns are elaborated below in detail.
There is substantial evidence that patent linkage delays generic competition, resulting in prolonged periods of high medicine prices borne by patients and the public health system. By delaying generic entry, such measures adversely impact the development of the generic sector and further exacerbateexisting challenges in access to affordable treatment. The proposed approach therefore risks undermining affordability and weakening Malaysia’s generic industry, particularly by exposing it to costly and strategic legal challenges initiated by patent-holding originator companies.




