1. Introduction
We urge the National Pharmaceutical Regulatory Agency (NPRA) to withdraw the proposed Guidelines on Implementation of Patent Linkage for Pharmaceutical Products in Malaysia and to undertake meaningful consultations with civil society organizations and patient advocacy organisations on the best way to approach implementation of Article 18.53 of the Comprehensive and Progressive Agreement for Trans-Pacific Partnership (CPTPP).
The proposed Guidelines are CPTPP-plus going well beyond what is required under Article 18.53 of the CPTPP and will have serious adverse consequences for both the generic industry and access to affordable pharmaceutical products in Malaysia.
High medicine prices are a persistent challenge in Malaysia placing a significant burden on both patients and the public healthcare system. As a result, patients face high out-of-pocket costs, while the government must allocate substantial resources to procure medicines, constraining public health spending. Thus, it is absolutely critical that NPRA's implementation of Article 18.53 of the CPTPP does not erect barriers to the early entry of generic competition. Early generic competition is essential to reducing prices and improving access, and any regulatory approach that delays it will only exacerbate existing affordability challenges.
Looking at 10 years of wholesale and retail prices of medicines in Malaysia (2012-2022) the trend is incremental price increase (median increase: 40% and 33.6%), or stable increase (median increase: 15% and 16%), but none showed a decrease. In fact, the recent 5-year trend shows that 66% of wholesale and 68% of retail medicines have become more expensive. The increase for wholesale and retail prices is highly correlated.
The Ministry of Health’s 2024 pooled procurement data shows that a switch from innovator (patented) to generic medicines produced significant savings for the Ministry: in all cases, the saving was averaged at RM6.8mil per medicine procurement. However, when the Ministry sticks to the same innovator medicine (and not a generic), there is a 1 in 4 chance that the Ministry must fork out more for the same procurement. In the other 75% of cases the savings the Ministry could gain per procurement of originator medicines is about 12% on average after price negotiations or bulk buying. This is small compared to the savings made by a switch to generics. Hence any delay in availability of a generic pharmaceutical product in the market has a huge cost to the government and the public. Timely registration of a generic product by NPRA is therefore crucial, together with a generic importer or local manufacturer that is ready to enter the market with the registration in hand.
In 2024, the Ministry of Health (MOH) spent RM823 million on 839 innovator medicines, of which 99% were imported. Even a modest delay in the entry of generics—affecting just 5% of these medicines—will have significant financial implications. Based on estimated switch savings, a delay of just 5% of generics could result in loss of savings of approximately RM285.6 million, equivalent to about 8.2% of MOH’s total annual expenditure on medicine procurement.
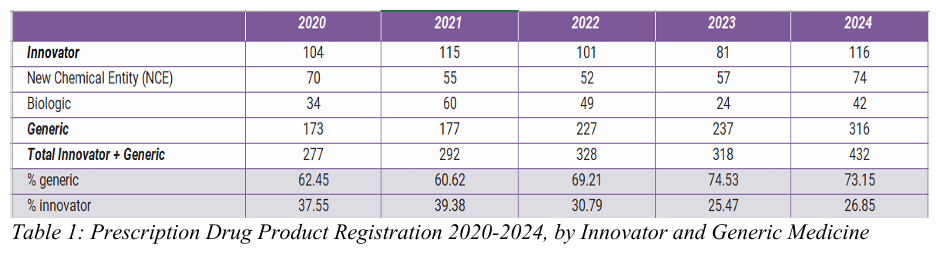 Over the five-year period between 2020 and 2024, the number and percentage of generic products approved by the NPRA has increased (See Table 1). In 2024, 316 generic prescription drugs were registered, accounting for 73.15% of the total. The progress and gains made under the National Generic Medicines Framework, which was developed based on the Malaysian National Medicines Policy and favours the use of generic medicines, will be reversed if the proposed Guidelines are adopted.
Over the five-year period between 2020 and 2024, the number and percentage of generic products approved by the NPRA has increased (See Table 1). In 2024, 316 generic prescription drugs were registered, accounting for 73.15% of the total. The progress and gains made under the National Generic Medicines Framework, which was developed based on the Malaysian National Medicines Policy and favours the use of generic medicines, will be reversed if the proposed Guidelines are adopted.
The Malaysian government has also identified the generic pharmaceutical industry as a strategic priority under the New Industrial Master Plan (NIMP) 2030. According to the NIMP, the Malaysian generic industry has grown significantly over the past decade, reaching 10 per cent growth of total local medicine sales between 2010 and 2021. The Plan sets out an ambition for Malaysia to maintain its position as a first mover in generics while expanding export capacity. More broadly, it envisions strengthening the industry to improve the affordability and accessibility of medicines, enhance national self-sufficiency, support local economic development, and reduce the pharmaceutical trade deficit. The NIMP pharmaceutical sector plan (2023) identifies evergreening patents as “significant challenge” of patents: “In Malaysia, the Patent Act does not exclude second medical use patent for pharmaceutical products. This can lead to monopolies and high prices for essential medicines. … Some companies may patent a new formulation with slight modifications of existing drugs. This allows the companies to extend its monopoly on the products and prevent generic competition.”
In this context, the proposed Guidelines are deeply concerning. In particular, the proposed Guidelines: (i) are CPTPP-plus; (ii) legitimises and promotes patent evergreening; (iii) its proposed patent listing is open to abuse; (iv) will encourage frivolous litigation against generic companies and create a chilling effect on generic market entry; (v) undermines the effective use of flexibilities in the WTO Agreement on Trade-related Aspects of Intellectual Property Rights (TRIPS); (vi) violates established principles of law; (vii) exceeds NPRA’s legal mandate and (viii) introduces legal uncertainty. These concerns are elaborated below in detail.
There is substantial evidence that patent linkage delays generic competition, resulting in prolonged periods of high medicine prices borne by patients and the public health system. By delaying generic entry, such measures adversely impact the development of the generic sector and further exacerbateexisting challenges in access to affordable treatment. The proposed approach therefore risks undermining affordability and weakening Malaysia’s generic industry, particularly by exposing it to costly and strategic legal challenges initiated by patent-holding originator companies.
2. Proposed Guidelines are CPTPP-plus
NPRA has proposed guidelines on implementation of patent linkage on the basis that this is a requirement under Article 18.53 of the CPTPP. But the CPTPP does not explicitly mention patent linkage (PL). In any case, PL is a TRIPS-plus measure and there is no one-size-fits-all approach to implementing PL even if a country decides to do it.
Article 18.53 of CPTPP provides two options for implementation. Of the two options, Article 18.53.2 is the worse option for access to medicines. There is no need to implement that option. The minimum required by Article 18.53.1 is a notification system and availability of judicial remedies.
NPRA has ignored this fact. Instead, NPRA’s proposed Guidelines suggests establishing 4 categories, requiring PRH NDP (i.e. the originator) and the PRH generic applicants to provide a list of patents and pending patents, imposing a 45-day notification period during which NPRA withholds registration decisions, and providing for an automatic 12-month suspension of the regulatory approval process if the patent holder initiates legal action. Further, the Guidelines proposes an expansive scope that includes secondary patents that will merely encourage “evergreening” of patents in Malaysia. None of these elements are required under the CPTPP. Notably, New Zealand and the United Kingdom—both CPTPP parties— with Gross National Income per capita 4 times larger than Malaysia have made clear that compliance with Article 18.53 of CPTPP does not necessitate such measures.
In particular, the 45-day notification period followed by an automatic 12-month suspension is unjustified and unnecessary. Section 60 of the Malaysian Patents Act 1983 (amended 2022) recognizes the possibility of the patent holder to apply for an injunction including for situations when the patent holder can prove imminent infringement. Such remedies are provided by Malaysia’s judicial system and can be obtained rapidly within 48 hours. There is no need for NPRA to do anything. CPTPP also only requires notice to be provided to the patent holder. But the proposed Guidelines require category 4 applicants to provide 2 notices, one to the patent owner or patent licensee and another to the product registration holder of the NDP.
The proposed Guidelines also extend to “For Export Only (FEO) products” which is not required by the CPTPP. The title of Article 18.53 of CPTPP is “Measures relating to the marketing of certain pharmaceutical products” and the sub-paragraphs are about marketing of the pharmaceutical product. In the case of FEO products, there is no marketing in Malaysia. Moreover, patents are territorial in nature, meaning that even if a pharmaceutical product may have some patents (valid or weak patents) in Malaysia, the same product may not have any patents in the country to which the product is exported. There is thus no legal basis for applying the proposed Guidelines to FEO products.
Against the above background, it is evident that NPRA’s proposal goes far beyond what is minimally required to implement Article 18.53 of CPTPP and risks serious negative consequences for generic competition and access to affordable pharmaceutical products in Malaysia.
We emphasise that any implementation of Article 18.53 of the CPTPP should be confined strictly to what is required under Article 18.53.1. In practice, this would only necessitate NPRA providing a simple public notice—such as publishing, on its website, information that a marketing approval application has been filed, shortly before approval is granted. Malaysian law already provides the necessary legal remedies under the Patents Act and the Rules of Court, and no additional regulatory measures are required to implement Article 18.53 CPTPP.
3. Proposed Guidelines Promotes Patent Evergreening
Patent evergreening is a common strategy used by pharmaceutical patent holders to extend market exclusivity beyond the standard 20-year term by securing additional patents on minor modifications to existing medicines, such as new uses, formulations, dosages, or delivery methods. These follow-on, or “secondary,” patents typically provide little or no meaningful therapeutic benefit. Instead, they are primarily pursued by multinational pharmaceutical companies to prolong monopolies, delay generic entry, and sustain high prices to maximise profits. In Malaysia, the absence of sufficiently rigorous patent examination by MyIPO has enabled the widespread use of such practices, contributing to delayed generic competition and persistently elevated medicine prices. As mentioned above this is a challenge identified in the NIMP pharmaceutical sector plan.
An often-cited example of patent evergreening is the lopinavir and ritonavir combination (LPV/r), a second line treatment for HIV. In Malaysia, although no primary patents existed on lopinavir or ritonavir compounds, secondary product patents on crystalline polymorphs, heat-stable formulations enabled AbbVie the patent holder company to maintain a prolonged monopoly and charge high prices. As a result, Malaysia remained dependent on purchasing Kaletra, the originator’s brand-name version of LPV/r. In 2018, the cost of the originator product (200 mg/50 mg) was approximately RM 3,103 per patient per year (around US$735 at the time), while generic versions were available for less than US$300 per patient per year.
Similarly, in Malaysia, the compound patent targeting cancer drug nilotinib expired in July 2023, but the patent grant of a nilotinib method of treatment has extended the patent duration until November 2030. In contrast, in India, except for the compound patent, the rest of the patent claims were either rejected or withdrawn. Thus, in India, with generic competition the nilotinib tablets cost around RM3 per tablet, while prices in Malaysia are between RM55 to RM70 per tablet, i.e. more than 18 times the price in India.
The proposed Guidelines legitimise patent evergreening, i.e. an abuse of the patent system. Section 4.4 of the proposed Guidelines allows patent linkage for product patents claiming active pharmaceutical ingredients, formulations, polymorphs, salts, esters, dosage forms, dosing regimens and approved medical use indications. These types of patents are almost exactly the list of what patent scholars call “secondary patents” – patents that pharmaceutical companies file after filing the original compound patent, to extend their monopoly. Empirical research shows that these types of secondary patents add on an average 6 to 7 extra years of patent life beyond the original compound patent and are disproportionately filed for best-selling pharmaceutical products. Despite this, NPRA has sought to recognize such frivolous patents, for the purpose of patent linkage.
When such secondary patents are linked to the marketing approval process, as proposed in the Guidelines, they create significant barriers to generic entry—even where no actual patent infringement exists. For example, under Category 3, secondary patents listed by the originator (PRH NDP) can effectively extend the perceived patent expiry date on an ongoing basis. Under Category 4, these same patents can be used to trigger legal proceedings including frivolous legal action against generic companies, resulting in suspension of the regulatory process.
Moreover, the proposed Guidelines permit PRH NDP (originator companies) to continuously update and expand their patent lists as new patents are granted. By recognising and incorporating secondary patents into the patent linkage mechanism, and allowing their ongoing addition, NPRA is effectively encouraging patent evergreening practices and facilitating the use of legal actions to delay or block generic competition in Malaysia.
Fixed dose combinations (FDCs): A number of important products for treatment are FDCs. For e.g. FDC for antiretroviral (ARV) include dolutegravir/lamivudine (Dovato), dolutegravir/rilpivirine (Juluca) and bictagrevir/emtricitabine/TAF (Biktarvy). The clearest example is TLD (tenofovir/lamivudine/dolutegravir) which WHO has recommended as the preferred first-line regimen for adults and adolescents and MOH is planning to accelerate transition to TLD. Similarly, a large number of treatments for non-communicable diseases such as cardiovascular and diabetes are FDCs.
Patents on FDCs are usually considered to be frivolous as they are essentially combinations of known compounds and thus lacking in novelty and inventive step. And when such patents are challenged through advocacy or opposition proceedings, the patent holder may abandon the patents. For example, patents sought over rifapentine and isoniazid combinations (used to treat latent tuberculosis infection) were withdrawn globally by Sanofi, once patent oppositions were filed in India and in other countries. Another example is the combivir patent, a fixed-dose combination ARV medication used to treat HIV/AIDS. It contains two ARV medications, lamivudine and zidovudine, and was used together with other antiretrovirals. It was first introduced to the market in 1997 and marked a major breakthrough as the first fixed-dose combination therapy for people living with HIV. In Malaysia, combivir was patented and the cost of originator combivir was very high. By issuing a government use license in 2003, Malaysia accessed affordable generic versions and the average cost of treatment per patient per month dropped dramatically—from US$315 to US$58, a reduction of approximately 81%. Following global protests against GSK’s frivolous patents over combivir, the company eventually withdrew its patent claims for the combination worldwide, including in Malaysia.
The proposed Guidelines seems to extend to FDCs as the definition of “pharmaceutical products” speaks of “combination of substances” and Section 4.4 includes “formulation” product patents. Given that such patents are known to be used strategically by patent holders to extend their market exclusivity without corresponding therapeutic advances, their inclusion raises serious concerns about patent misuse to delay generic entry.
In sum, the scope of patents eligible for linkage is excessively broad, extending far beyond primary patents on new chemical entities (NCEs) to encompass a wide range of secondary patents. This approach effectively legitimises and entrenches patent evergreening, creating a clear pathway for patent holders to game the system. By allowing such patents to trigger regulatory barriers, the linkage mechanism becomes a tool to prolong market exclusivity, delay generic entry, and sustain high prices at the expense of patients, our public health system, and timely access to affordable medicines.
4. Patent Listing proposed by the Guidelines Open to Abuse
Patent listing is not a prerequisite for implementing Article 18.53.1 of the CPTPP. Experience from countries that have incorporated patent listing into their linkage regimes shows that such systems are prone to abuse. Patent holders have used listing strategically to include irrelevant or weak patents, creating unnecessary barriers for generic entrants and triggering stays of the regulatory process. Rather than safeguarding legitimate rights, patent listing will be used to deter competition and delay the entry of more affordable medicines.
In the US, improper listing has become a major concern. Its Food and Drug Administration (FDA) acts in a purely administrative capacity, taking patent declarations at face value. But the US Federal Trade Commission (FTC) has found many instances of improper patent listing aimed at delaying generic competition. In 2023, the FTC issued a policy statement explicitly stating that “Brand drug manufacturers may be harming generic competition through the improper listing of patents in the Food and Drug Administration’s (“FDA”) Approved Drug Products with Therapeutic Equivalence Evaluations, known as the “Orange Book.” It further notes in its policy statement that: “Improper Orange Book patent listings may disincentivize investments in developing a competing product and increase the risk of delayed generic and follow-on product entry, reducing patient access to more affordable prescription drugs and increasing costs to the healthcare system. Given the enormous profit margins of many branded drugs, even small delays in generic competition can generate substantial additional profits for brand companies at the expense of patients.”
The Chair of the FTC has also added that “[i]mproper patent listings in the Orange Book illegitimately delay or lock out generic manufacturers from entering the market, depriving Americans of access to lower-cost medicines and drug products”. In 2025, the FTC challenged more than 200 improper patent listings.
In June 2005, a report of the United States House of Representatives Committee on Government Reform Minority Staff, highlighted the impact of patent linkage on access to generic medicines and noted that “even the FDA does not have adequate expertise or resources to review the applicability of patents, and it has been unable to prevent abuses of the system by patent holders that have led to delays in the availability of generic drugs”.
An example of improper listing:

Similar challenges exist in Canada that also requires patent listing as part of its linkage mechanism. The Canadian government and its Supreme Court recognised that the patent linkage system was abused by the originator companies by continually adding irrelevant and frivolous patents to the list of patents notified to the regulatory agency, which were then used to block the entry of generic competition for a long time at huge cost to the Canadian health system.This is more so when the scope of the linkage system is expansive and includes secondary patents as suggested in the NPRA Guidelines.
We reiterate that patent listing is not a requirement for implementing Article 18.53.1 of CPTPP. In any case, there is significant uncertainty in the Guidelines as to how the listing will operate.
According to the Guidelines, the Category 2 applicants (i.e. the patent holders of the originator product) will list the patents granted and pending patent applications that are applicable to their product. Generic companies falling within Category 3 (i.e. generic applicants that will market the generic product once the patent expires) are also expected to list all active patents. In all likelihood, the list of patents by Category 2 applicants will differ from the list of patents of Category 3 applicants. Since the Guidelines allow patent evergreening, it can be expected that the Category 2 applicants will continue adding patents to the list and extending the patent expiry date. However, if the Category 3 applicants are of the view that relevant patents have expired, but Category 2 applicants have a separate list of patents, what are the implications? NPRA is rightly indicating that it will not be involved in any patent dispute; however there is legal uncertainty on how any difference in the listing of patents will be addressed and the effect on the registration of the generic product.
Section 6.2.2 of the Guidelines expects the generic company to “ensure that the product is marketed only after the patent of the corresponding NDP has expired”. This suggest that NPRA expects generic companies to be guided by the list of patents provided by the Category 2 applicants, when determining the patent expiry date. This approach will motivate the originator to continue listing frivolous, irrelevant patents as a deterrent to generics entering the market.
5. Proposed Guidelines Will Encourage Frivolous Litigation Against Generic Companies and Create a Chilling Effect on the Generic Industry
The proposed Guidelines pose a serious threat to local generic companies and the development of Malaysia’s generic industry. In particular, Category 4 exposes generic companies to frivolous litigation by patent-holding originator firms. Even where there is no genuine patent infringement, originator companies have a strong incentive to initiate legal proceedings, as doing so will automatically trigger a suspension of the registration process. They may then prolong these proceedings to benefit from the full 12-month delay. Such delays enable originators to maintain market exclusivity, sustain high prices, and maximise profits. In effect, NPRA’s proposal creates a structural incentive for increased litigation against generic companies in Malaysia and will significantly delay the entry of affordable generic products.
The proposed Guidelines also undermine a very important business strategy of generic companies i.e. to “launch at risk” where they are aware that the remaining patents are weak and/or will not be infringed.
Most generic companies in Malaysia are small and medium enterprises (SMEs) that lack the financial capacity to withstand repeated or prolonged legal challenges. As a result, the threat of litigation alone— even in the absence of any actual patent infringement—can deter these companies from seeking marketing authorisation. This creates a chilling effect, discouraging generics from entering the market as early as possible, not due to legal merit, but to avoid the risk and cost of frivolous lawsuits.
The British Generic Manufacturers Association (BGMA) presenting evidence to the Parliament in the United Kingdom on the potential impact of mandatorily notifying the originator, when the generic companies launch as risk highlights that: “[l]egal proceedings may be triggered by prior notification and commonly delay generics getting to the market by several years. This pushes some generic launches past the originator loss of exclusivity date. Moreover, some generic companies may be reluctant to file an MA [marketing approval] in the patent term fearing the front-loaded cost of legal action”.
The BGMA further adds that “If multiple generics are likely to come to the market at the same time, the first to decide to enter the market does so for everyone, necessarily incurring the time, expense and litigation risk of the legal proceedings. The share of the generic market for that product ultimately available to the generic litigant may be less valuable than the cost of litigation” adding as well that “reluctance to be first to file MA applications would significantly delay generic market entry for some patent protected medicines. Moreover, it would make it riskier and more expensive for a generic competitor to consider launching where a weak patent exists, thus deterring some from developing a product to bring to market at all.”
The link between patent linkage mechanisms and increase of patent litigation is undeniable. Studies in both the United States and Korea have shown that more patents have been filed after the coming into force of patent linkage provisions with a surge in patent litigation. The increase in patent litigation is detrimental to access to affordable medicine. The increase in litigation costs is a heavy burden especially for SMEs, impacting their ability to bring generics into the market.
In the US, the Federal Trade Commission (FTC) also found that, in 73% of the cases in which a decision was reached on the merits, the generic company was successful in invalidating the patent, clear evidence that most of the legal proceedings initiated by the patent holder were frivolous, mainly aimed at delaying generic competition. As highlighted above, even if the patents are weak/invalid, Malaysian generic companies, which are mostly SMEs will be deterred by the costs of litigation.
Interestingly in the US, the FTC also found that in several cases, the issue of patent validity was never determined as the patent holder and the generic company concluded settlements that delayed generic market entry. “Pay-to-delay” patent settlements, a by-product of the linkage system are now the subject of further FTC scrutiny as they keep more affordable generic versions off the US market, costing consumers and taxpayers US$3.5 billion in higher drug costs every year.
In Canada, linkage has also created a significant backlog in the Canadian Federal Court system. In 2008, there was a team of approximately 30 Federal Court judges devoting some or all of their time to about 350 separate drug patent cases and “The Supreme Court of Canada has, on multiple occasions, held that the automatic stay issued to patentees under the NOC Regulations is an 'extraordinary' remedy, not available to patentees in any industry outside of the pharmaceutical industry.”
6. Proposed Guidelines Undermine Use of TRIPS Flexibilities
A major concern with the proposed guidelines is the undermining of TRIPS flexibilities.
(a) Nullifies the benefit of “Bolar exception”
The purpose of the Bolar exception in Section 37(1A) of the Patents Act 1983 is to allow a generic company to apply for registration before the expiration of the patent term so that the generic can enter the market as soon as the patent expires. However, NPRA’s proposed introduction of a complicated system for implementing Article 18.53 of the CPTPP, means that the benefit of this exception will be nullified. Generic companies which believe that their products do not infringe existing patents, will fall within Category 4, and will face the possibility of legal action, with their registration process automatically suspended.
(b) Undermines use of TRIPS flexibilities to curb patent evergreening.
An important flexibility available under the TRIPS Agreement is the right to define the patentability criteria in a way that limits patent evergreening, a major concern in Malaysia. Yet the proposed Guidelines entrench this practice. By effectively recognising secondary patents within the regulatory framework, NPRA’s approach also undermines incentives for MyIPO to fully utilise TRIPS flexibilities, including adopting stricter patentability standards and more rigorous examination practices.
7. The proposed Guidelines Violate Established Principles of Law
Article 18.53(1)(b) and (c) of the CPTPP requires that legal remedies are available to the patent holder and that it has adequate time and opportunity to seek such remedies. These requirements are already met in Malaysia by provisions in its Patents Act as well as in its Rules of Court 2012.
Section 59 of the Patents Act provides that a patent holder may initiate infringement proceedings where there is likelihood of “imminent infringement”, while Section 60 provides that if the patent holder “proves imminent infringement the Court shall grant an injunction to prevent infringement and any other legal remedy”.
Order 29 of the Rules of Court 2012 set outs the provisions for applying for interim injunction. Order 29 in fact allows for ex parte (from one party) injunctions whereby injunctions can be granted within 24 to 48 hours to the applicant where the legal conditions are met. Where an injunction is granted ex parte, it is strictly limited in duration: the order automatically lapses 21 days from the date it is granted unless it is continued or confirmed by the court, and an inter-partes (between parties) hearing must be fixed within 14 days of the ex parte order. This time bound structure ensures that while the affected party has been given a prompt opportunity to be heard, the remedy is provisional and the court is able to quickly reassess the correctness and necessity of the injunction, thereby reinforcing proportionality and fairness of the system.
Critically, in granting an ex-parte injunction, the court must first be satisfied that there is a serious question to be tried, and if so, must then consider whether damages would be an adequate remedy and, if not, where the balance of convenience lies. In the Court of Appeal case of Motor Sports International Ltd (Servants or Agents or Agents at Federal Territory of Labuan) & Ors v Delcont (M) Sdn Bhd [1996] 3 CLJ 483; [1996] 2 MLJ 605, His Lordship Gopal Sri Ram JCA (as he then was) succinctly held as follows:
"The provisions of O 29 r 2A were introduced by amendment in order to ensure that ex parte injunctions of any sort were not granted willy-nilly but only in cases where they were truly called for. In order to ensure that the policy behind the introduction of r 2A is not defeated, high courts must demand strict compliance with its terms. More so, when the relief applied for is in the nature of a Mareva or an Anton Piller type of injunction because of the incalculable harm and damage that may be caused to a defendant by the grant of either of these orders."
The standards for granting interim measures is reflected in the High Court´s judgment in Lim Choong Huat & Ors v Syntlz Enterprise Sdn bhd & Ors [2010] 1 CLJ 860, wherein His Lordship reminded that the Patents Act 1983 “makes the court the ultimate adjudicator to decide on the validity of the patent; however, pure registration of the patent alone does not give any certainty to the patentee of his right as a valid patent holder… it cannot be said that prima facie the patentee has the exclusive right of making, using or selling etc of his product. The Act only gives the true and first inventor a limited monopoly, as a reward for the invention. The question whether he is the true and first inventor can seldom be determined at the application stage at the patent office”.
In this context, NPRA’s proposal to grant automatic stay on the regulatory approval process upon receiving evidence of legal proceedings undermines the existing procedure under Order 29 of the Rules of Court 2012 and runs counter to established Malaysian law. The courts have consistently treated interim injunctions as exceptional remedies, subject to strict legal and procedural safeguards, precisely to avoid premature deprivation of a person/entity’s legitimate rights without full inter-partes scrutiny. By contrast, a Category‑4‑based regulatory stay would impose an effective de facto bar on market entry to the detriment of the defendant, based solely on mere initiation of legal proceedings, bypassing the requisite judicial scrutiny required under Order 29. Such an action amounts to a extrajudicial quasi-injunctive mechanism that is beyond NPRA´s jurisdiction and power.
8. Proposed Guidelines are Beyond the Authority of NPRA
The proposed Guidelines are issued by the Senior Director of Pharmaceutical Services “pursuant to Regulation 8 of the Control of Drugs and Cosmetics Regulations 1984”. Section 2 of the Guidelines asserts that the Guidelines ensure “regulatory neutrality”. Thereafter Section 6.3.2 of the Guidelines states that NPRA will “refrain from assessing, interpreting or adjudicating the validity, scope, ownership, enforceability or alleged infringement of any patent”. It further adds in Section 6.3.3 that “All matters relating to patent rights, including validity or infringement, shall remain within the exclusive jurisdiction of the courts or other competent adjudicating authorities”.
While claiming NPRA will not be involved in any patent-related matters, the proposed Guidelines require Category 2, 3 and 4 applicants to list their patents. For Category 3 applicants, the Guidelines permit registration but requires the generic PRH to ensure that the generic product is marketed only after expiry of the corresponding patent. This essentially amounts to patent enforcement via regulatory conditions, even though the Patents Act recognises patent enforcement to be the responsibility of the patent holder and done using court processes. Section 37(1A) of the Patents Act reinforces that regulatory submission acts are exempt, so the regulatory approval pathway itself should not be transformed into a patent-control/linked mechanism.
For Category 4 applicants, the Guidelines state that NPRA shall commence a 45-day notification period during which period no regulatory approval decision will be granted, followed by an automatic 12 month suspension of the regulatory approval process, if legal proceedings are initiated by the originator/patent holder.
These stay mechanisms effectively turn NPRA into a patent-enforcement gatekeeper even though patent-related matters fall under the authority of MyIPO, and patent enforcement matters within the jurisdiction of the court system. It is also highlighted above (in part 7) that NPRA’s proposal to impose a stay on regulatory approval undermines the Rules of Court 2012 and established Malaysian law, and amounts to a extrajudicial quasi-injunctive mechanism that is beyond NPRA´s jurisdiction and power.
NPRA says it will only check procedural compliance and will not assess patent validity, scope or infringement. However, its procedural verification is tied to mandatory regulatory consequences that affect approval timing and market entry. This is not “regulatory neutrality”. On the contrary, that combination makes NPRA the operational gateway for private patent enforcement which exceeds its health-regulatory remit.
From this perspective, NPRA’s proposed Guidelines are clearly beyond its statutory role under the Sale of Drugs Act 1952 (SDA) and the Control of Drugs and Cosmetics Regulations 1984 which regulate product registration on public health grounds, principally quality, safety and efficacy. The Guideline’s 45-day notification period and 12-month suspension period are not tied to any deficiency in product quality, safety or efficacy; they are made available only to facilitate enforcement of patents which is a private right.
Under the Patents Act 1983, the proper forum for scrutiny of infringement allegations is the court. Section 59(2) recognises imminent infringement actions and section 60(2) allows the patent holder to seek injunctions from the court, where the legal system can weigh the merits and courts regularly require an undertaking as to damages which serves as a promise by the patent holder to compensate the defendant for any losses suffered if it is later determined that the injunction should not have been granted. The undertaking mitigates the risk of unfairness to the generic applicant if the patent is not ultimately upheld. NPRA has no comparable adjudicative mandate, and no clear statutory power to create de facto injunctive relief by suspending regulatory approvals at great cost to the generic applicant.
In our view, the proposed Guidelines cannot be justified under Regulation 8 or NPRA’s parent legislation, the SDA as NPRA is using its health-registration powers to facilitate private patent enforcement. NPRA does not have the legal authority to suspend regulatory decisions as a patent enforcement remedy.
9. Proposed Guidelines Create Uncertainty
The proposed Guidelines will generate significant uncertainty.
(a) Section 5.1.6.8 states that “the notice shall be acknowledged by the patent owner or patent licensee and PRH NDP within seven (7) days”. But what happens if the patent owner or patent licensee delays in providing acknowledgement or does not provide any acknowledgement?
(b) Section 5.2.2 states that “Failure to notify NPRA within the stipulated timeframe may result in appropriate administrative action under the applicable regulatory framework”. There is no clarity on what the administrative actions will be.
(c)The Guidelines do not provide clarity on whether the patent information provided by the Category 2 applicants will be made publicly available even though in Section 2 of the Guidelines, it is claimed that it will be a “transparent administrative mechanism”. Yet, there is no clarity which aspects will be transparent.
(d) The Guidelines might also have an impact on fast-track registration pathways presently provided by NPRA e.g. a generic approved under priority review procedure presently expects to be approved within 100 working days and the generic facilitated registration pathway under ASEAN Joint Assessment Procedure, has an approval timeframe of 60 days. There is little clarity on how this will be managed.
(e) There is also the possibility that a PRH NDP or the patent holder/licensee may seek to trigger back to-back suspension of the regulatory process, by continuously updating its patent list (which the Guidelines allow). For example, towards the end of the first 12-month suspension, a PRH NDP or the patent holder/licensee adds more patents to the list, triggering 45 day notification and another 12-month suspension. Such as scenario has happened in other countries implementing similar systems; how will this scenario be managed by NPRA?
10. Conclusion: The impact of the proposed Guidelines on the generic industry, generic competition & access to affordable pharmaceutical products will be devastating
Section 2 of the proposed Guidelines claims that the mechanism will be “safeguarding access to medicines” while Section 3 of the proposed Guidelines claims that patent linkage is a “regulatory mechanism that aims to balance incentives for innovation and access to affordable medicines.” Both assertions are unfounded. In practice, there is substantial evidence that patent linkage delays generic entry, resulting in prolonged periods of high medicine prices borne by patients and the public health system. Rather than achieving a balance, the proposed approach risks undermining access to affordable medicines. The Guidelines undermine the objectives set out in NIMP 2030. It will weaken Malaysia’s generic industry, which is likely to struggle with the additional barriers to market entry—particularly the increased risk of costly and strategic legal challenges initiated by patent-holding originator companies. Further, the progress achieved under the National Generic Medicines Framework— grounded in the Malaysian National Medicines Policy and designed to promote the use of generic medicines—risks being undone by the proposed Guidelines.
Specifically on patent linkage, the UN Special Rapporteur on the right to health has highlighted that it “would affect the entry of generic medicines” adding that by “delaying the process of granting marketing approval, patent linkage provides patent holders with additional opportunities to prolong their monopoly rights and delays the entry of generic medicines into the market”. The Special Rapporteur also stressed that “[p]atent linkages, by not allowing the registration of generic versions of patented drugs can also adversely impact the early working exception, which ensures the immediate entry of generic competition after the expiry of the patent. Similarly, refusal to register also creates uncertainty in relation to compulsory licences”.
Patent linkage also increases pharmaceutical imports of the originator’s product, as the domestic generic industry will be prevented from supplying, thus affecting Malaysia’s trade balance of payments (i.e. more imports than exports) and employment if the local generic industry is unable to grow. In a global environment of unstable global supply chains a weakened local generic industry will also undermine Malaysia’s national pharmaceutical supply security.
In conclusion, we reiterate our call and request NPRA to withdraw the proposed Guidelines on Implementation of Patent Linkage for Pharmaceutical Products in Malaysia and to undertake meaningful consultations with civil society organizations and patient advocacy organisations on the best way to approach implementation of Article 18.53 of the CPTPP.
Contact:
Mohideen Abdul Kader
President, Consumers Association of Penang
This email address is being protected from spambots. You need JavaScript enabled to view it.
Datuk Indrani Thuraisingham
Vice President and Legal Advisor, Federation of Malaysian Consumers Associations (FOMCA) This email address is being protected from spambots. You need JavaScript enabled to view it.
Chung Han Yang
Deputy Executive Director Malaysian AIDS Council (MAC)
This email address is being protected from spambots. You need JavaScript enabled to view it.
Dr Murallitharan M.,
Managing Director, National Cancer Society of Malaysia
This email address is being protected from spambots. You need JavaScript enabled to view it.
Chee Yoke Ling Executive Director,
Third World Network
This email address is being protected from spambots. You need JavaScript enabled to view it.